NOx’s Effect
- Photochemical Smog
- Greenhouse Effect
- Acidification of soil by acid rain
NO generated during the combustion process is rapidly oxidized to NO2 when released into the atmosphere, but NO2 is reduced back to NO by photolysis in the atmosphere. During the process, free oxygen radical reacts with oxygen molecules in the atmosphere to form O3. This reaction accelerates the reaction when the hydrocarbon (most VOC materials) are present in the atmosphere, (See Figure 1) light NO2 → NO + O, O + O2 → O3 The generated O3 is present in the atmosphere for a long time and causes photochemical smog and greenhouse effect. Is known to be a toxic substance that has a direct impact on the environment.
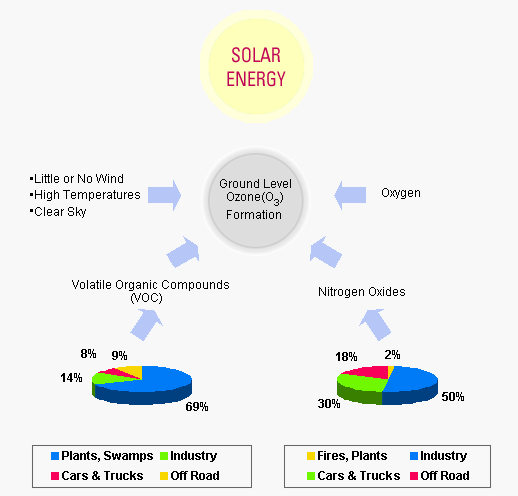
Figure 1 Distribution of the NOx Emissions from All Sources
(Source: Special Report a Summary of NOx Reduction Technologies
The Texas Institute for Advancement of Chemical Technology, 2000)
Atmospheric NO2 reacts with hydroxyl radicals to form nitric acid. The nitric acid produced is absorbed in water or precipitated into the soil with nitric acid particles themselves, which acidifies the surrounding soil.
NO2 + OH → HNO3
In addition, NO2 and NO are absorbed in rainwater and produce nitric acid, which affects soil acidification in the surrounding area.
2NO2 + H2O → HNO3 + HNO2
NO2 + NO + H2O → 2HNO2
In addition, it has been reported that O3 is formed in the atmosphere and acidification of soil through various routes.

